understanding acid rain
When Gene Likens discovered that rainwater collected in the Hubbard Brook Experimental Forest
in 1963 had a pH of 3.7, this began a long series of efforts to understand
why this rain was so acidic, how this acidity compared to other places on earth,
and what the source of this acidity was. It took more than a decade for researchers
to establish that this rainwater acidity was not natural (rain in the most remote regions
of the Earth typically is 100x less acidic than samples collected from Hubbard Brook in
the 1960s) and that the source of this acidity in the rain falling in central New Hampshire
was from the smokestacks of coal fired power plants in the Ohio River valley.
(read more about this discovery here)
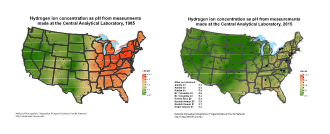
FIGURE 1 – Hydrogen ion concentrations as pH from measurements made by the National Atmospheric Deposition Program in a) 1985 and b) 2015. Note that each dot represents a precipitation monitoring station. Maps for all years available at http://nadp.isws.illinois.edu/maplib
Not Everywhere:
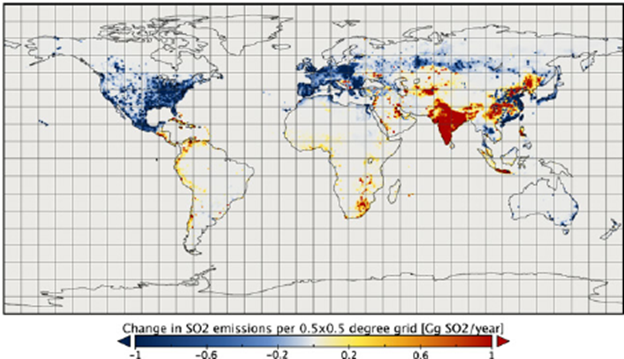
FIGURE 2. A map of the change in global sulfur dioxide (SO2) emissions between 2005 and 2010. Note the decline throughout North American and Europe and the widespread increase in southeast Asia (this figure is from Klimont et al. 2013 and is available at this link.
Not Quite:
What to look for in this data story
RESOURCES TO LEARN MORE
Free internet resources
Books
Key Papers